Also, eat all the leafy greens you can get: seaweed snacks, Mom’s collard greens, kale krunchies, spinach salad, you name it. It’s amazing nerve food.
For antioxidant powerhouses, look for dark-red and dark-blue fruits: pomegranates, blueberries, red wine, chocolate (though some CRPS people have to avoid that for its nerve effects), mangosteen (my favorite fruit), cranberries, and so on.
Stay smart. Stay loose. Keep going.
For medical people — clinical takeaways:
Most treatment standards, particularly for CRPS, are based on science that’s over a decade old. They shouldn’t be changed blithely but they can certainly be improved. There is plenty of room for that.
The following points are intended as additions to the standards you follow for CRPS, as they are good guidelines for mitochondrial and neurologic support in a system compromised by CRPS.
- After any limb surgery, give Vitamin C 500 mg, QD or BID, for a couple weeks beforehand and 30-50 days after — or to metabolic tolerance, if that’s too much. Use a food-associated form for best uptake. This one intervention will reduce the risk of developing CRPS by 80%, according to the best current data.
- We assume your patients are taking an adequate multivitamin and are eating plenty of greens, dark fruits, and wholesome proteins. So make sure they are. Direct them to food bank, food stamps or other food assistance as needed. Give recipes. (No kidding.) 2 benefits: better antioxidant uptake if taken with antioxidant-rich food, and increasing the patient’s own sense of agency/participation improves pain and affect. (If you don’t believe in multivitamins, then get out of the supermarket/pharmacy and get some real ones.)
- Stress the antioxidant vitamins. In acute CRPS, give water-soluble antioxidant vitamins in 1-3x the doses you’d give a healthy person. Give fat-soluble antioxidants (A, D, E) up to 2x normal, testing levels as indicated. Consider vitamin K inj.
- In cold/chronic CRPS, give water-soluble antioxidant vitamins in 3-5x the doses you’d give a healthy person (start at 2x and work up). Give fat-soluble antioxidants (A, D, E) up to 2-4x normal, testing levels as indicated; consider weekly mega-dose D (as used in AIDS.) Give vitamin K inj. Check serum or urine levels as indicated, especially as we develop absorption disorders.
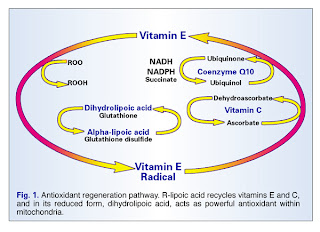
- Give “uber-antioxidants” like ubiquinone (co-Q 10), N-acetylcysteine, or glutathione. There are indications that these can provide substantial benefit — though again, not normally curative of chronic CRPS. They are impressive, especially for mitochondrial-dysfunction issues.
These ranges are empirical; if you can find the funding to do the science to develop more reliable ranges for this population, so much the better.
Adequate tissue oxygenation and perfusion can return substantial function and significantly reduce pharmacologic burden. Patients can demonstrate this, even where the data have not been published and peer reviewed. Therefore, use antioxidants rigorously and intelligently.
- Between the cortisol and systemic oxidative stresses, it can’t hurt and it will help something. You’ll see a distinct improvement in affect, activity, motivation and well-being when the dose is optimized, even if it can’t be expected to be curative. Making your patient’s life more bearable is an essential part of your job.
- Let’s say this together, everyone: statistics mean nothing in the case of the individual. Accepted, standardized medicine is what you start with, but, when your case is taking you out to the margins, you go to the margins, because that’s where your success is most likely to await.
Keep in mind that doctors are not the only scientists interested in the human body. Be prepared to look into other disciplines for leads when your own offers no good options.
Try Nursing, PT, Nutrition, Therapeutic Massage — you’ll realize that nobody knows more about soft tissue’s functional physiology in vivo than therapeutic massage science, and if nothing else, the exercise in intellectual flexibility might do you good.
The accepted style is very different, but the info they have is tremendous.
Forward-looking thoughts:
- Consider infusing vitamin K into CRPS-damaged tissues. I would love to see studies on that.
- Figure out how to deliver antioxidants in a targeted way. (Now! Please!) This would be a good way to save a lot of lives and end tons of misery.
… And for all curious people …
Let’s go back to mitochondria in reproduction. Kind of in an X-rated way, figuratively speaking.
We know that women have two X chromosomes. The Y chromosome is a stubby little object with hardly any data to use, unless you’re into color-blindness or hemophilia; this means women have quantities of extra data, which can have even more devastating effects (as in, Down syndrome.) So how to handle the extra genes?
Pick one. Simple as that.
Shortly after conception, when the cells are just dividing like mad and haven’t decided what to be yet, every single cell turns off one of its two X chromosomes; each of that cell’s daughter cells inactivates the same X chromosome. As the cells continue to multiply, then fill out, fold, bend around, and specialize, to become a whole, separate being, it means that X-linked traits appear in a mottled pattern throughout the body, as the two sets of daughter cells continue reproducing and passing on their particular X-activations. Isn’t that curious?
As an especially decorative instance, cats’ hair color is an X-linked trait:
Cool, huh? Love her accent, too.
But this fact brings me to a serious question about mitochondrial disease. If mitochondria are sex-linked, is there a relationship between the X chromosome and mitochondrial expression? It seems improbable that there wouldn’t be, because mitochondria reside inside the cell, and the cell’s action is determined by the genes within it. The mitochondria had to have developed a special relationship with the X’s in the 23rd chromosomal pair, after all those millenia.
It’s generally accepted that mitochondrial diseases are due to toxification or to complex, multigenetic issues. Ok, fine. But what about mitochondrial vulnerabilities that don’t become pathologic until they are damaged in some other way? To what degree is toxification an issue related to X-activation? In other words, is mitochnodrial vulnerability related to vulnerabilities in the active X chromosome?
Is there a patchy characteristic to the early stages of mitochondrial destruction? — You know, the early stages of rare disorders, the time when it’s impossible to get a diagnosis because the doctors are all so busy chasing their own tails around your irrational symptoms and their own ignorance.
Is that initial “mottled” activity one reason why these diseases are so damn weird?